Episode 056: Is Elon Musk’s Neuralink a Science Fiction Horror Show or the Salvation of Humanity?
In this week’s episode, I sat down with Dr. Michael Cummings, renowned psychiatrist and long-time friend of The Psychiatry & Psychotherapy Podcast at Patton State Hospital in San Bernardino, CA for a mind-blowing discussion on past advances in neuroscience and new developments in neurotechnology news.
By listening to this episode, you can earn 1 Psychiatry CME Credits.
Other Places to listen: iTunes, Spotify
Article Authors: Victoria Burghart, Valerie Pooudomsak, David Puder, MD
There are no conflicts of interest for this episode.
How Neuralink, Elon Musk, Brain-Machine Interfaces, And Deep Brain Stimulators May Change Psychiatry Forever.
Neuralink’s official presentation cites three main goals driving their creation of a Brain Machine Interface:
To Understand and Treat Brain and Spinal Disorders
To Preserve and Enhance the Brain
To Create a Well-aligned [better] Future
One of the most practical applications for the device is allowing those with quadriplegia to interact with a digital environment or mobile device, improving their quality of life. In theory, this could also give the human brain, with its biological processing, access to the power and efficiency of high-speed digital processing. Could Brain Machine Interfaces be the next giant leap forward in the evolution of humanity? Elon Musk certainly thinks so.
One of the more controversial components of the Neuralink presentation was Musk’s inclusion of his beliefs about the future of humanity and artificial intelligence. During the press release he stated one of his goals was to create the ability to achieve a “full symbiosis with artificial intelligence,” essentially removing the “existential threat of AI” which he believes will one day “leave us behind” (Neuralink, 2019). This goal has been met with a bit more skepticism, especially by the national media, than the medical applications of Neuralink’s BMI. Forbes describes it as “a bit more fantastical” than the company’s primary goal of treating brain disorders (Knapp, 2019). Other publications have been far less kind, such as The Atlantic, which published its coverage of the Neuralink press release with the title: “Elon Musk’s Next Wild Promise: If someone is going to revolutionize what it means to be human, do we want it to be a tech titan?” (Mull, 2019). Although the New York Times surmised that “one of the biggest challenges may be for his scientists to match his grand vision,” (Markoff, 2019), it serves as a good example of what most major media outlets have chosen to do: stick to the facts.
Dr. Cummings and I (Dr. Puder) believe that these technological advances may help those who suffer with severe mental illness. There are still patients who don’t respond to therapy, medications, and treatments like ECT. It is likely that Neuralink’s plans for a BMI are not impossible, as the same thing is already being done on a small scale with brain stimulators. The hope for BMI is that this type of treatment could be done in a more detailed and nuanced way. In this next section, we will explore potential therapeutic targets for some of the mental illnesses that are the most challenging to treat.
A BRIEF INTRODUCTION TO NEURAL PROCESSING:
The human brain is the most advanced biological processing system on the planet; equivalent to a one-trillion bit per second digital processor. There are an estimated 86 billion neurons in the human brain, with up to 10,000 connections to other neurons, forming a collection of 1,000 trillion synaptic connections. This massive aggregate of neurons and synaptic connections allows the brain to store between 1 and 1,000 terabytes of memory. For comparison, the U.S. Library of Congress contains around 19 million works, collectively storing 10 terabytes of data. The scientific community has been studying the brain for decades, and while our body of knowledge regarding its function has increased exponentially, exploration of one of humanity’s greatest frontiers is still underway.
Biological information processing in the brain is a complex non-linear system, founded on electrochemistry in and around the neuron. Some outside stimulus, such as a sensory cell or another neuron, changes the electrochemical properties of the area around a neuron, generating an electrical depolarization called an “action potential”. The action potential ripples from the first neuron to other neurons that it shares synaptic connections with. This chain reaction carries the information into the brain where it is sorted into the area that processes that specific type of information, then the processed information is used for a variety of purposes: to make memories, inform decisions, or react immediately. To borrow Dr. Cummings’ excellent analogy, biological processing is a bit like an anthill with different divisions that have different tasks and all work simultaneously, while none of these individual divisions are “in charge” at any given time. Digital processing is more linear in nature: a command is given to the processor and then the processor uses that information to execute a specific response based on its design.
The complexity of the brain’s biological processing, while amazing and intricate, does have a downside: how do you fix the system when something goes wrong? Malfunctioning neural networks, imbalances in levels of functioning between areas of the brain, or loss of neurons are all causes of psychiatric and neurological disease. Treating this dysfunction, especially when severe, is a massive challenge for psychiatrists, psychologists, and therapists. From a biological perspective, there are two broad categories of treatment that change neuron function: (1) pharmacologic and (2) direct modification of neuron function. Pharmacologic treatment uses medication to modify the chemical environment around neurons to change their function. Direct modification of neuron function is more invasive and uses application of electrical current or a magnetic field to modulate neuron function.
ARE WE USING DIRECT MODIFICATION OF NEURON FUNCTION TO TREAT PATIENTS TODAY?
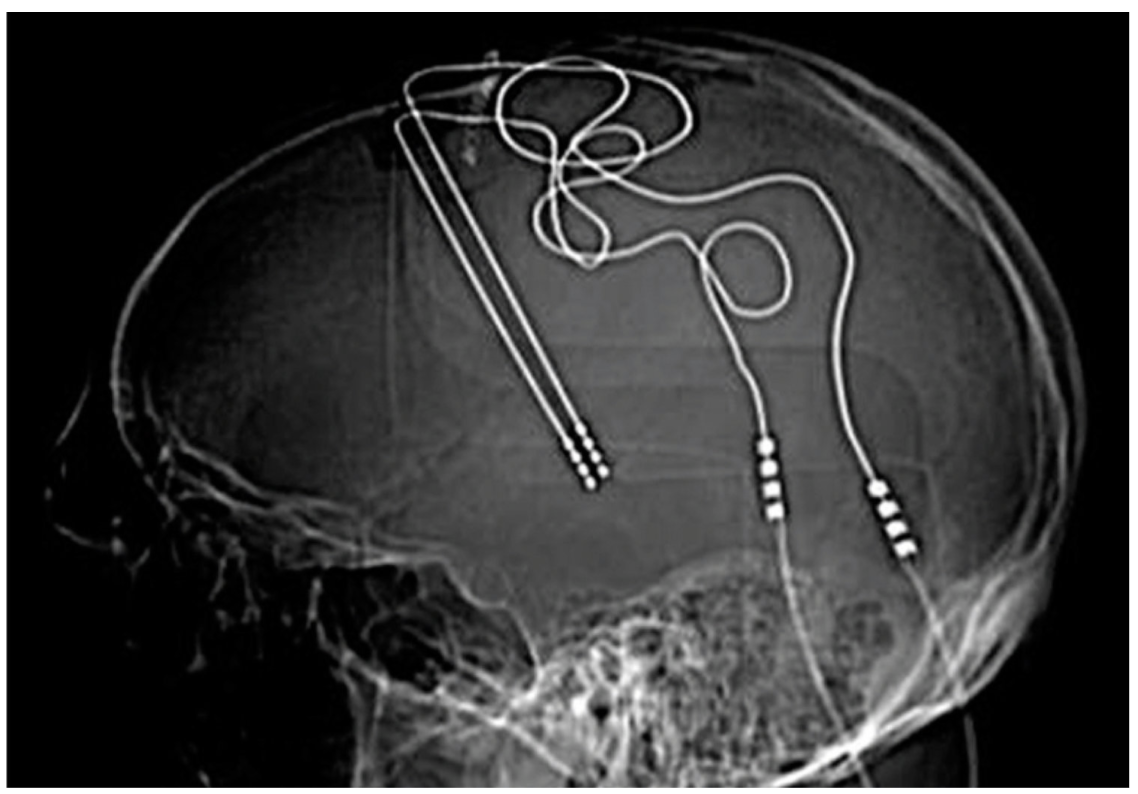
Deep Brain Stimulation (DBS) uses application of electrical current to change neuron function via electrodes directly implanted into the brain. DBS is commonly used for movement disorders like Parkinson’s disease and in some cases of severe, treatment refractory depression. Electrodes for DBS treatment require a neurosurgical procedure with the patient awake, under local anesthesia, so they can give feedback to the surgeon as they test each electrode. Two implants, with a total of 5-20 electrodes, are placed at target sites in each hemisphere of the brain. During the procedure, the patient tells the surgeon if movement is easier, if they feel different, etc. and the surgeon can select which 2-4 electrodes should be used to produce the most beneficial effect. The surgeon then places connecting wires from the implants inside the brain out through the skull and under the skin to a pacemaker-like device on the chest wall; this device interfaces with a handheld device in the physician’s office, where they can change the intensity and duration of electrical stimulation.
Image Source: Chen, 2019.
DBS has been dramatically successful in about ⅓ of patients with severe, medication refractory symptoms. It is possible that DBS may fail to relieve symptoms, due to issues with electrode placement or ineffective stimulation, in addition to all of the risks that accompany neurosurgery such as infection. While all of these potential side effects are possible, catastrophic failure of DBS is relatively rare.
DBS for depression typically targets the anterior portion of the limbic system, especially the circuit of Papez. One of the many theories of refractory major depression is an imbalance between the limbic circuits on the dominant and inferior lobe. If the dominant hemisphere’s limbic system is underactive, symptoms of major depression may arise.
Rapid Transcranial Magnetic Stimulation (rTMS) uses electromagnets to create a rapidly changing magnetic field that induces an electrical current in the brain tissue underneath the magnetic coil. This creates a similar effect to DBS with direct stimulation of neurons, without implanting a device, but lacks the potential for the constant stimulation necessary to treat some conditions such as Parkinsons and other movement disorders. TMS is further limited by its inability to target small or deep areas of the brain, but lacks the risk associated with neurosurgery.
ELON MUSK & NEURALINK’S PROGRESS: HOW CLOSE ARE WE TO A BRAIN-MACHINE INTERFACE?
Neuralink is a neuroscience research and development company that was founded in 2016 by Elon Musk and other developers with the ultimate goal of creating a Brain-Machine Interface (BMI). On July 16th, 2019 Musk and Company live-streamed a press release detailing their work to-date which coincided with the release of a journal article about their work on creating a BMI.
If you would like to watch the full press release or read the full article, you can find both here:
An Integrated Brain-Machine Interface Platform with Thousands of Channels, Musk, E. (2019).
Neuralink has spent the last two years building a complete BMI and surgical robot to inplant the BMI. Creating a device with the potential to function for a long period of time, while being both effective and safe required Neuralink to engineer components for their interface from scratch. The main components of the device are: the threads, the hermetic substrate, and the high-density electronics.
One thread is composed of a miniscule strand of flexible polymer filament and up to 128 electrodes. Each thread is about 5 microns thick, or approximately 20x smaller than a human hair. This size is comparable to the average human neuron, which measures between 3 microns in diameter at the axon and 9 microns in diameter at the dendrite (Lennie, 2003). The threads are created through a 3D printing process and packaged as a sterile unit with the hermetic substrate and electronics.
Hermetic substrate is the term for the sterile packaging around a component of a medical device that cannot react with the biological environment, in this case, the capsule around the electronics. Neuralink has developed a material called a “monolithic thin film substrate” which is manufactured with the threads to create a single unit. This avoids the technical issues that are associated with the historical method of creating a hermetic substrate: using heat to place a conductive material through holes in a non-reactive, inflexible substrate which is prone to cracking, thus losing its sterile seal.
The high-density electronics created to do all the processing for the BMI were designed to be brain-friendly: tiny, low power, highly modifiable, and able to collect data from a large number of electrodes. The first model chip measures 4mm by 5mm, which is equivalent to the diameter of a standard plastic drinking straw. The N1 chip can process data from 1,124 individual electrodes, convert the data from analog to digital, and recognize the spikes that represent action potentials from neurons. Each electrode takes 20,000 readings per second, producing 200 Megabytes of data per second per implant. The algorithms programmed into the chip then compress the data for export and further analysis about 200x, all within about 900 nanoseconds. The chip can also stimulate neurons on up to 64 electrodes at a time. In theory the device could “read” motor information, which could be used to control various devices such as mobile phones and “write” sensory information back into the brain.
Neuralink scientists are also working on interpreting the data gathered from neuralink devices. This includes developing algorithms to detect and interpret patterns of neurons firing during movement. During the Neuralink press conference Elon Musk leaked information concerning their recent primate studies with the BMI containing the N1 chip, stating that “a monkey has been able to control a computer with its brain, just fyi...” (Neuralink, 2019). Neuralink developers postulate that the applications for this technology will only be limited by the technical abilities of the system itself and the speed at which it can be developed.
Neuralink plans to conduct its first clinical trial in human subjects in 2020. The study will be limited to participants with quadriplegia due to cervical spinal cord injury. Each patient will receive four N1 sensors, placed in the motor cortex, supplementary motor area, and the dorsal premotor cortex, with closed-loop feedback into the primary somatosensory cortex. Patients should be able to use the system at home, using a wireless interface called a “pod”.
Potential Applications For The Brain Machine Interface In Psychiatry
The future for Brain Modulation in MDD, OCD and Schizophrenia…
Depression
Complex BMIs like Neuralink have great potential as a therapy for treatment-resistant depression. This type of depression is the most severe, long-term form of depression. These patients haven’t responded to multiple types of medications, often spending over 5 years with their psychiatrist “trialing” the different options. These people have suffered from years of depression and deal with significant suicidality. According to the Hamilton Rating Scale for Depression (HAM-D), severe depression symptoms are characterized by:
Reporting virtually only feeling sad, hopeless, helpless, or worthless, in their spontaneous verbal and nonverbal communications
Hears accusatory or denunciatory voices and/or experiences threatening visual hallucinations
Attempts to commit suicide
Complains of nightly difficulty falling asleep
Complains of waking during the night
Complains of being unable to fall asleep again if they get out of bed
They have stopped working because of their present illness. If hospitalized, they don’t engage in any activities
Slowness of thought and speech; impaired ability to concentrate; decreased motor activity - to the point of stupor
Agitation - hand-wringing, nail-biting, biting of lips
Mental anxiety - expresses their fears without being questioned
Physical anxiety - often incapacitating, hyperventilation or sighing, urinary frequency, sweating
Somatic GI symptoms - difficulty eating without urging by others. Requests or requires laxatives or medications for bowels or medications for GI symptoms
General somatic symptoms - any clear-cut physical symptoms
Severe sexual symptoms, such as loss of libido or menstrual disturbances
Hypochondriacal delusions
Loss of weight - either definite weight loss according to patient history or more than 2 lb. of loss per week while hospitalized
Complete lack of insight - denies being ill at all
There have been several studies addressing the efficacy of DBS for use for those with severe, treatment-resident depression. Improvement of depression symptoms has been shown in four areas of the brain: the subgenual anterior cingulate cortex, the ventral capsule & ventral striatum, and the nucleus accumbens (Drokbisz & Bambroská, 2019). Imaging studies conducted in TMS patients have also implicated the subgenual anterior cingulate cortex as part of dysfunctional neural networks found in depression patients (Phillip, et al. 2018) . In general, when treated with DBS at one of these four locations ⅓ of patient experienced complete remission of depression symptoms, ⅓ showed improvements in symptoms, and ⅓ received zero benefits from treatment (Drokbisz & Bambroská, 2019). More complex BMIs could provide the chance to more closely monitor neural networks, fine-tune regulation of dysfunctional groups of neurons, and customize individual treatments.
Schizophrenia
Direct neuromodulation via systems like Neuralink or DBS have the potential to become useful for treating severe schizophrenia when someone’s symptoms don’t respond to Clozapine, the gold-standard for treatment-resistant Schizophrenia. (To listen to Dr. Puder and Dr. Cummings episode about Clozapine being used for treatment resistant Schizophrenia click here) Symptoms of severe Schizophrenia include:
Persistent hallucinations
Delusions
Disorganization of speech and behavior such as tangential or circumstantial speech, derailment and neologisms
Severe cognitive impairment, such as decreased processing speed, issues with both verbal and visual learning and memory
Difficulty controlling mood or anxiety
Suicidality
To listen to Dr. Puder and Dr. Arianna Cunningham discuss the clinical manifestations of schizophrenia, click here.
A 2016 study conducted in rat models of schizophrenia found that implanted and activated DBS probes in the brain appears to have improvement schizophrenia- like symptoms (Klein, et al. 2013). Potential targets for direct intervention include (1) the hippocampus, which has been implicated in excessive dopamine release leading to psychosis, (2) the ventral striatum, part of the polysynaptic pathway required for the hippocampus to release extra dopamine, and (3) the associative striatum (which includes the caudate nucleus), another potentially important locus of abnormal dopamine release (Mikell, Saurabh, & Sameer, 2016). The dysfunction of the arcuate fasciculus between Wernicke’s area and Broca’s area is another brain area thought to contribute to the formation of auditory hallucinations that could potentially be a target for neuromodulation.
Obsessive-Compulsive Disorder
Severe treatment resistant OCD has great potential for personalized care via direct modification of neural function. Symptoms of severe OCD include:
Persistent obsessions or compulsions
Obsession: recurrent, persistent thoughts, urges, or images that are intrusive and unwanted, causing anxiety or distress that the individual attempts to ignore, suppress or neutralize with another thought or an action
Compulsions: repetitive behaviors or mental acts that the individual feels driven to complete in response to an obsession or to rules that must be rigidly followed. These mental acts and behaviors are completed in order to reduce anxiety or distress, but may not be directly connected to the stressor (obsession)
Obsessions or compulsions consume a significant portion of the individuals time, to the point where they cannot function well in society
When asked about where we might target OCD in the future, Dr. Cummings explained the dorsal striatum (which includes the caudate nucleus) is where we store pre-programed motor behaviors, the things we don’t want to think about and don’t want to dedicate cortical time to (including grooming and orienting behaviors). When the caudate nucleus thinks we need to check, it will generate a signal that goes from the anterior caudate nucleus to the orbital cortex; this triggers a behavioral response to go turn the stove off or make sure the door is locked. This behavior generates a negative feedback from the cortex back through the globus pallidus and then up to the caudate nucleus to turn that behavior off. In OCD, the “off” side, or the inhibitory side does not work right. Then caudate keeps perseverating, or orienting, or grooming signal causing these people to get stuck in rituals. Electronically, if we were to boost the “off” signal when the negative circuit is activated it might help fix the illness.
Although the caudate circuit (as described above) was considered, a recent review discussed the role of the dorsal anterior cingulate cortex (dACC); one of the dACC’s main functions is to help the brain ignore unimportant information and inhibit rapid “automatic” responses in favor of slower, more “reasoned” responses (McGovern, Sameer, & Sheth 2017). This makes the dACC a strong candidate for direct neuromodulation. Studies of DBS efficacy in the treatment of OCD have targeted several other areas including: the anterior limb of the internal capsule, the ventral capsule/ventral striatum, the nucleus accumbens, the subthalamic nucleus, and the inferior thalamic peduncle, all of which had an overall response rate around 50% despite variability between studies (Koning et al. 2011). While DBS and future neuromodulating treatments require further research, they have potential for significant improvements in patient outcomes.
Source: McGovern, Sameer, & Sheth 2017
Race To The Frontier: Other Neuroscience Developers
Kernel
Founded by Bryan Johnson in 2016 with the intent to develop neuroprosthetics that can enhance cognition. Most recently the company has produced a neuromodulator which can read a very high capacity of neural signals. (Richardson, 2017)
IBM
Study in February 2019 used the TUH EEG Seizure Corpus database obtained from scalp EEGs to explore the possibility of machine learning algorithms to classify of different seizure types. They achieved a weighted F1 score of 0.907 for their algorithms, indicating a high level of accuracy. (Roy et al, 2019)
John Hopkins
Study in February 2019 suggests that electrocorticography recordings of the sensorimotor cortex could support the development of a whole limb brain-machine interface which could control the movements of multiple joints. (Thomas et al, 2019)
MIT
Study in 2017 used EEG to detect real-time error-related potentials in the brain which were then decoded and used to control a robot in a binary selection task. (Salazar-Gomez, 2017)
Researchers at UCSF used high-density electrocorticography recordings, filtered in real-time recordings to isolate high gamma frequency range signals representing multiple units of sensorimotor neurons in conjunction with the context of participants speech to develop algorithms capable of almost instantaneously identifying participants’ responses to a standard set of questions with predefined answered sets. They reached an accuracy of 61% for produced speech and 76% for perceived speech. (Moses et al, 2019)
BrainGate
Three participants with quadriplegia used an implanted BrainGate brain-machine interface in the motor cortex to record real-time neuronal activity; this was routed to a Bluetooth interface that allowed them to use apps on tablet devices such as web browsing, emailing, and sending messages. (Nuyujukian et al, 2018)
Will we be able to record dreams?
EMG has been used to record nerve impulses during REM sleep with the data uploaded into a simulator that created these body movements. (Oldis, 2017)
Current research is ongoing with implanted electrodes in brain surgery patients with the goal of understanding how we make decisions when we are awake and asleep. (Cerf, 2018)
fMRI has been used to record activity in the visual cortex in conjunction with participants’ dream recollections to map out patterns that could be used to predict what we dream about. (Horikawa et al, 2013)
Brain Chips
In this study, two monkeys with paralyzing spinal injuries were able to walk following implantation of brain chips in the motor cortex and stimulators in the lumbar spinal cord. (Courtine et al, 2016)
Researchers in this study implanted microelectrodes into the motor cortex of monkeys and decoded their cortical activity, which was then used to control wheelchair movements. With training, the monkeys were able to use their minds alone to move the wheelchair towards a reward grape. (Nicolelis et al, 2016)
Microelectrodes that were implanted into the motor cortex recorded cortical activity and were decoded, allowing monkeys to use their minds to control a prosthetic arm to successfully feed themselves (Schwartz, 2008)
Where Do We Go From Here?
It’s a very exciting time to be alive with all of these new developments in neurotechnology. Potential applications in the field of mental health could provide hope for effective treatment of severe mental illness. There is an astronomical amount of research that needs to be conducted before these applications can be realized, but it appears that the speed of research in the field is picking up the pace. As with all developing fields of research, the ethics of use and clinical practice will still need to be examined to prevent every sci-fi enthusiast’s nightmare. The challenge of balancing caution and open-mindedness is inherent in the process of discovery. Devices like Neuralink have the capacity to change and shape the way we view and interact with the world. The real question is, will it change us or will we use this new tool to change the world?
“Those who cannot change their minds cannot change anything.” - George Bernard Shaw
Engage in the dialogue on Dr. Puder’s Instagram, Facebook, Twitter